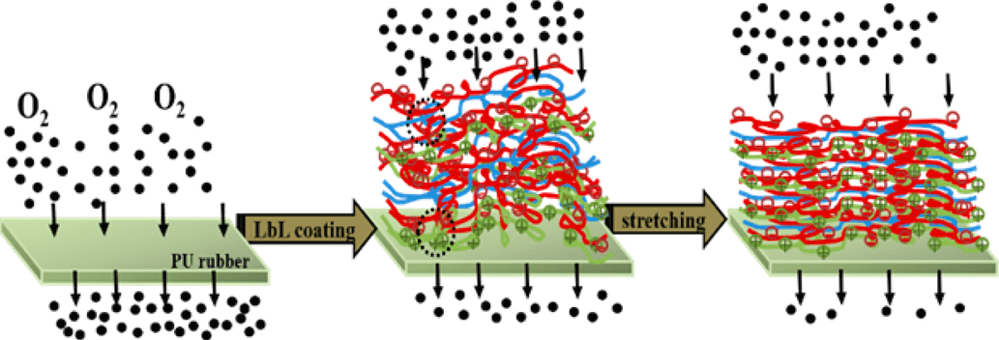
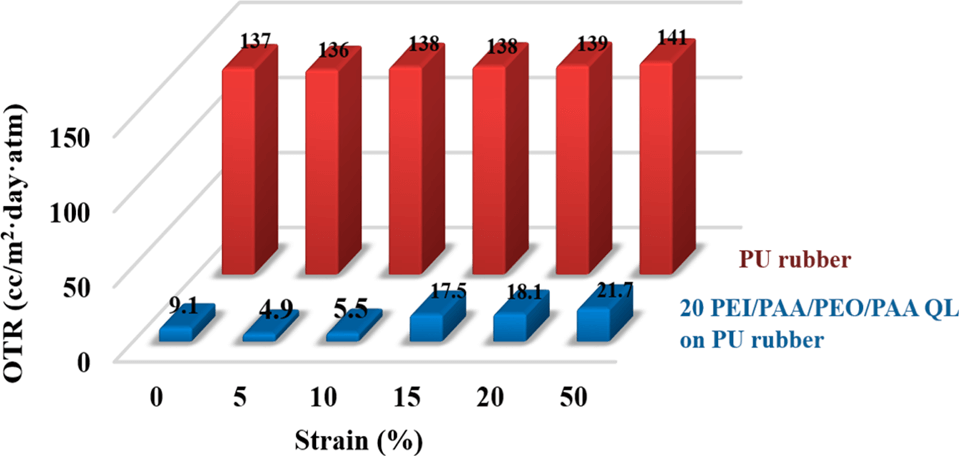
Ionically-bonded polyelectrolytes deposited layer-by-layer can achieve an oxygen transmission rate (OTR) below 0.005 cm3/m2 day atm (0% RH, 23°C), which exceeds the gas barrier of SiOx and metallized films. Although they are very flexible, these super barrier films often crack at 10% strain due to high stiffness. By using hydrogen bonding, “stretchable” assemblies can be achieved.

All-polymer hydrogen bonded LbL assemblies, such as poly(ethylene oxide) (PEO) and poly(acrylic acid) (PAA), or PEO and tannic acid (TA), can be stretched more than 20% with little or no loss of barrier. A 20 BL PEO/PAA thin film (367 nm thick) sustains its OTR at 100% strain (ACS Macro Letters 2014). Another study finds that replacing PAA with TA, a hydrogen bonding donor with a smaller molecular weight, makes thin films even more stretchable. A 40 BL PEO/TA assembly (344 nm thick) preserves the OTR when exposed to high intensity cyclic strain (20X at 100% strain) (ACS Appl. Mater. Interfaces 2015). Although extraordinary stretchability is observed in these hydrogen-bonded thin films, the gas barrier is relatively low owing to the large free volume that acts as an oxygen molecule pathway. Better gas barrier, while maintaining stretchability, is achieved with a PEI/PAA/PEO/PAA quadlayer system. In this case, a combination of ionic and hydrogen bonding is employed. This study further improves the barrier of elastomeric materials (e.g. inflatable elastomers used in tires and seals) (Macromolecules 2015).